Naughty Noble Gases
- Oct 20, 2017
- 3 min read
Overview
Noble gases are a topic that have been often dismissed in schools, as all we learn about them are that they are unreactive. However, many of you will know that this statement is false. Many noble gases do indeed form compounds, despite the fact these compounds don’t exist naturally.

Noble gases can be sorted into 2 groups; Radon, Xenon Krypton and Argon, Neon, Helium. The first group, containing Rn, Xe, Kr are more reactive than the second group of Ar, Ne, He. Rn, Xe, Kr are large noble gases with a large atomic radius. This means that electrons in outer shells of these noble gases experience a weak attractive force from the nucleus of the atom, so a very electronegative element, such as fluorine, can snatch electrons from a large noble gas, thus forming a weak Noble-X bond.
Examples of noble gas compounds include XeF4, Kr2 etc.

Synthesis
Since noble gases a full outer shell electron configuration, they only form compounds with other species under extreme conditions, such as high pressure and/or temperature.
Xenon compounds

Xenon is known for forming multiple compounds, such as XeF2, XeF4, XeF6. The synthesis of XeF6 requires 120°C and 60atm. Synthesis of XeF6 (Xenon hexafluoride) follows the equation:
Xe + 3F2 -> XeF6
Figure SEQ Figure \* ARABIC 1
- Structure of XeF6
Xe + 3F2 -> XeF6
In a normal laboratory, these conditions can be achieved using a small industrial sized pressure reaction vessel. By mixing Xenon gas and Fluorine gas in a 1:3 mol ratio, XeF6 will form under the conditions stated. Once formed, XeF6 will sublimate into a yellow vapour if kept at 120°C.
Helium compounds
Since helium is such a tiny element, consisting of only 2 protons, 2 electrons and 2 neutrons, it rarely forms any stable compounds and requires ridiculous conditions to force any reactions. Recently, scientists managed to synthesize a stable Helium compound: Na2He. The exact equation for the synthesis of this compound is still unknown, but scientists reacted sodium clusters (Na8) with helium gas under immense pressure, 300 Gigapascals to be precise.
To achieve the conditions of 300 Gpa, scientists used a diamond anvil cell to synthesize Na2He. A diamond anvil cell is a piece of scientific apparatus used to synthesize compounds under extreme pressure and/or temperature.

Figure SEQ Figure \* ARABIC 2 - Structure of Na2He
The diamond anvil cell consists of 2 diamonds facing each other, with the tips of the diamond anvils roughly 100-250 microns apart. Usually, a medium of inert gas surrounds the matrix of the cell, such as neon or xenon. The unreactive medium distributes the pressure from the diamonds evenly across the entire sample.

Figure 3 - Diamond Anvil Cell
[endif]--When Na8 clusters and Helium gas are fired through the cell under 300 Gpa, Na2He will form. Figure 4 shows the rearrangement of electrons within the Na and He atoms when pressure increases form 100 Gpa to 300 Gpa. Na2He is formed as a stable electride solid. Electride compounds are compounds in which the electrons act as anions.
Krypton compounds
The most abundant compound of Krypton is KrF2. KrF2 is synthesized by the following equation:
Kr + F2 -> KrF2
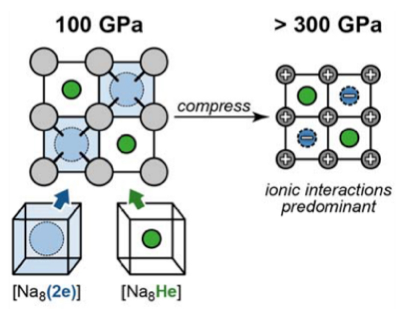
Figure 4 - The transformation
[endif]--Many methods are available for the synthesis of KrF2; methods include using a sophisticated particle accelerator (cyclotron), hot wire method etc. However, the photochemical method of synthesis of KrF2 is the most efficient and the easiest to carry out under normal laboratory conditions.
Firstly, samples of Krypton must be cooled to a solid by allowing a sample of Krypton gas to solidify in liquid nitrogen within a sealed fume cupboard. Secondly, samples of Fluorine gas must also be cooled to by liquid nitrogen to a liquid. Once Kr(s) and F(l) have formed, the samples are mixed in a clear, Pyrex container, with the container exposed to UV light via a UV lamp, set to 301nm. The yield of KrF2 should be roughly 510mgh, producing colourless crystals.
Neon compounds
Neon rarely forms stable compounds. One compound which does exist, however, is NeAuF (Neon gold fluoride). This compound is formed from a matrix isolation, which involves a guest material (neon gas) trapped within an unreactive host material matrix (AuF).

Figure 6 - Matrix isolation

Figure 5 - Apparatus for synthesis of NeAuF
[endif]--Synthesis of NeAuF involves the dilution of Ne(g) in gas phase with AuF. The reactants are fired through a beam and deposited on the window of the device, shown in Figure 5. Liquid Helium is pumped through the device, cooling the mixture to below the melting point of Neon. Once the sample solidifies, IR spectroscopy confirms the existence of this compound. Since this compound is extremely hard to synthesize and is unstable, scientists still do not fully understand the exact structure of this compound and types, distances and angles of bonds within the structure.
Conclusion
The field of noble chemistry is still developing and scientists uncover new findings every day. One day, we might even discover a bizarre fullerene structure of noble gases….
![endif]--![endif]--![endif]--![endif]--![endif]--![endif]--







Comments